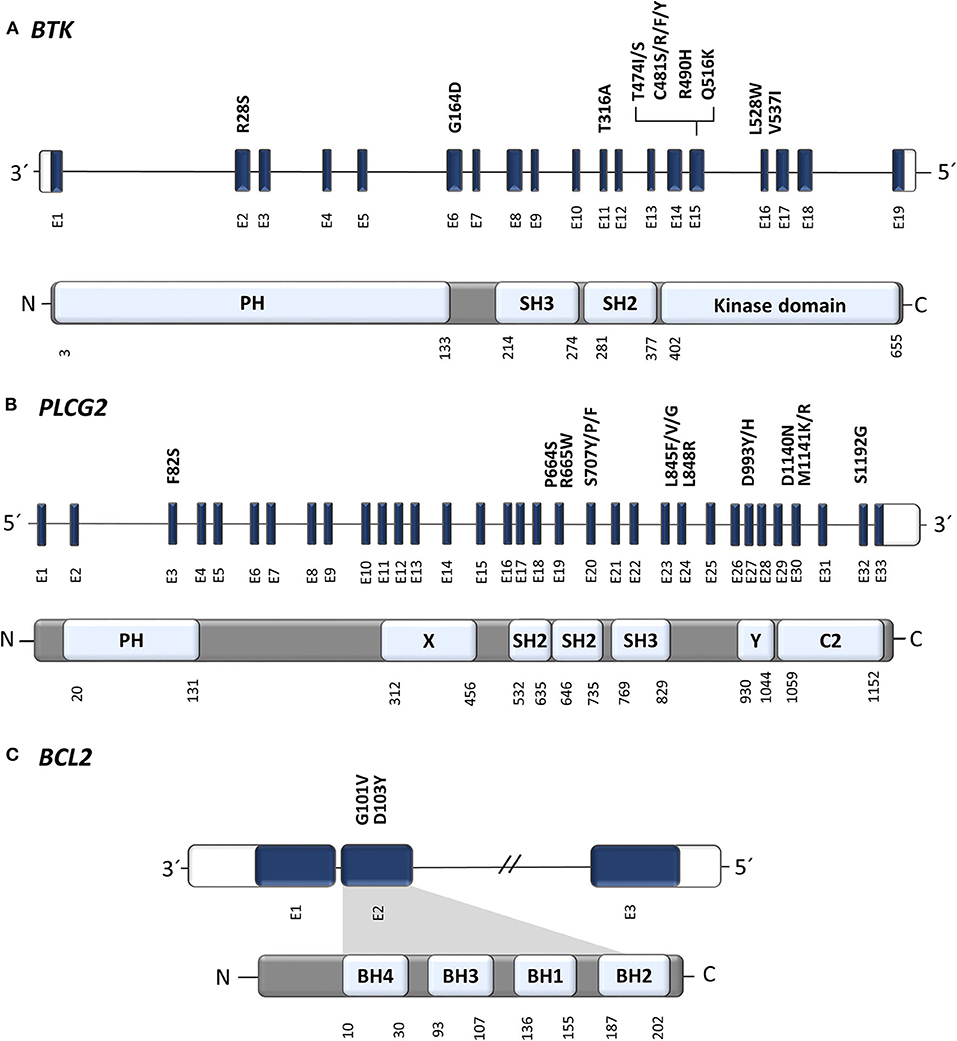
Consistent with increases in exposure, pBTK knockdown was observed. Preliminary PK data showed ARQ 531 exposure was close to dose proportional and the estimated plasma half-life generally ranged from 20-24 hours. The CLL patient with 29% tumor reduction had BTK C481S mutation. Three of 5 SD patients (1 follicular lymphoma, 1 DLBCL, 1 CLL) had 35%, 34% and 29% tumor reduction and 2 of them are ongoing on study treatment at 53 and 18 weeks. Among the 12 patients who have received at least 1 dose of study drug and have at least 1 post-treatment tumor measurement data, 5 achieved stable disease (SD) (1 follicular lymphoma, 1 DLBCL, 3 CLL) and 7 had progressive disease (6 CLL, 1 follicular lymphoma). No drug related serious TEAEs were reported. Drug related grade 3 or worse TEAEs included lipase increased and platelet count decreased in one patient (6.3%) each. Drug related TEAEs included diarrhea, nausea, vomiting, fatigue, pneumonia, amylase increased, lipase increased, neutrophil count decreased, platelet count decreased, decreased appetite, hypernatraemia, arthralgia, groin pain, dizziness, facial paralysis, headache, tremor and restlessness in one patient (6.3%) each. No dose limiting toxicities have been reported with ARQ 531. 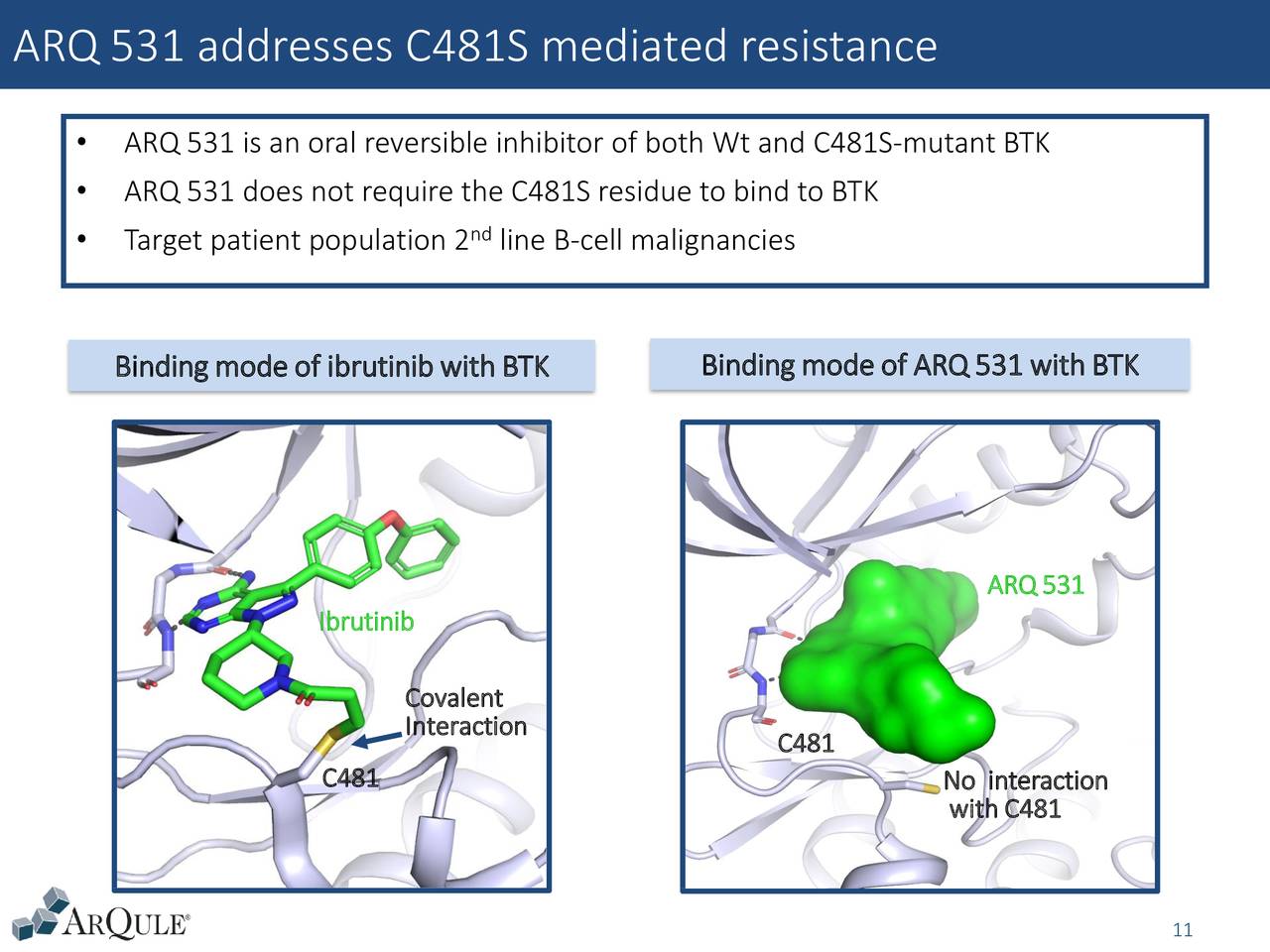
Results: A total of 16 patients enrolled (median age 65.5 years, male 93.8%, 2 DLBCL, 2 follicular lymphoma, 12 CLL/SLL, 5 median prior systemic regimens) and were treated at dose levels of 5, 10, 15, 20 and 30mg QD. Tumor responses were evaluated per disease specific guidelines. Treatment emergent adverse events (TEAEs) were assessed per NCI CTCAE v.4.03. Dose escalation was performed according to a 3+3 study design. Prior therapy must have included a BTK inhibitor, if FDA approved for their disease. Methods: This is a first in human, Phase 1 dose escalation study in patients with relapsed or refractory CLL/SLL, Waldenstrom's macroglobulinemia, or B-cell NHL who received at least 2 prior lines of systemic therapy. The secondary objectives are to assess the pharmacokinetic (PK) profile, pharmacodynamic (PD) activity, and preliminary evidence of anti-tumor activity of ARQ 531. Objectives: The primary objectives of the clinical study are to assess the safety and tolerability, and to determine the recommended Phase 2 dose and schedule of ARQ 531. ARQ 531 is a reversible ATP competitive inhibitor of BTK that inhibits ibrutinib-resistant BTK-C481S mutant CLL cells and has demonstrated antitumor activity in CLL, Richter's transformation, and DLBCL mouse models. Background: Aberrant activation of B-cell receptor (BCR) signaling is considered to be a major oncogenic mechanism that leads to the development and progression of multiple B-cell malignancies.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
